The Elemental Basics
 Figure 1- Hydrogen's place in the periodic table
Figure 1- Hydrogen's place in the periodic table
- Most abundant element in the universe; also makes up stars
- Known as an energy carrier
- "A secondary form of energy that has to be manufactured like electricity" [1]
- Doesn't occur naturally on Earth, like natural gas or oil
- "Carrier for storing and transporting energy from any one of a myriad of sources to where it may be utilized" [2]
- There is a large variety of methods to produce Hydrogen. Steam Reformation and Electrolysis are covered below.
- When used as a fuel, there is almost zero emissions.
- Best energy to weight ratio of any fuel (due to its density, only 7% that of air) but has a very poor energy to volume ratio, which creates storage problems.
Why do we need a new fuel source?
|
Production: Steam Reformation
The Present
Steam Reformation uses natural gas, which consists predominately of methane and is widely available, easy to handle, and currently cheap. Natural gas also minimizes the amount of carbon dioxide produced when it is reformed to produce hydrogen.
|
Pre- Treatment (#1 on figure 3): Water is softened and demineralized while the natural gas is desulphurised
Reforming & Steam Generation (#2 on figure 3): Steam is generated by boiling water. After being treated with a Nickel catalyst, Steam and methane are reacted at 900 degrees Celsius. This produces carbon monoxide and 3 units of hydrogen gas, the first reaction in figure 4. Water-gas Shift Reaction (#5 on figure 3): This step consists of four vessels with different absorbers that purifies the Hydrogen, represented by the four silver vessels in the top right of the picture. The overall reaction is the second reaction in figure 4. CO2 and H2 is produced from CO and Water. This step can yield up to 99.999% pure hydrogen. The last reaction in figure 4 is the combination of reaction one and two. Information from Towards Sustainable Road Transport by Ronald M. Dell, Patrick T. Moseley, David A. J. Rand, 2014 |
Problems with Steam Reformation
- Carbon Dioxide is still produced, so environmentally, there is not much net loss of emissions
- The conversion rate for feedstock to Hydrogen is good at 67-73%, however more progress is needed
- Prohibitive costs for applications like road transport
- Storage of hydrogen still remains a problem
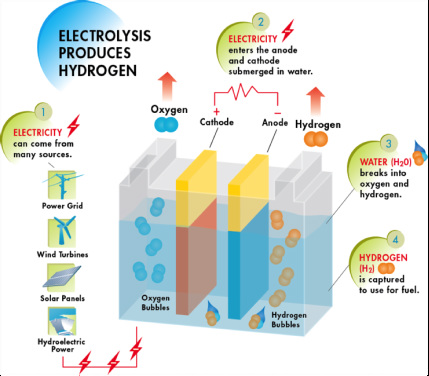
Production: Electrolysis
The Future
|
Electrolysis is the process of using electricity to split water into its constituents- hydrogen and oxygen.
The process is fairly simple. Figure 5 describes the process completely. Water is placed in a container with two electrodes, a cathode and an anode. Electricity is applied to the cathode and anode which begins to break down the water. Hydrogen begins to bubble up near the anode while Oxygen bubbles up at the cathode. Fresh water of high purity, important because salts would accumulate and clog the electrodes, is needed. Problems with Electrolysis
|
Information taken, unless explicitly stated, from Tomorrow's Energy by Peter Hoffman, 2012. Chapter 4: Producing Hydrogen from Water, Natural Gas, and Green Plants
|
Conclusions Regarding Steam Reformation and Electrolysis
|
Currently, 95% of Hydrogen is made through Steam Reformation and 4% by Electrolysis. The main reason for this is because Steam Reformation is incredibly cheap compared to electrolysis and much more efficient in terms of Hydrogen Production. However, in order to reduce emissions in every facet of energy production, Electrolysis is the method of the future. Steam Reformation produces Carbon Dioxide, which obviously still harms the environment. In order for Electroylsis to become a viable option, green methods of energy production (like the ones in the picture to the must be used to break apart the water molecule. According to studies by Argonne National Laboratory, 3,000,000–3,500,000 BTUs of fossil energy are used for the production of 1,000,000 BTUs of hydrogen through fossil energy-based water electrolysis. [4] This demonstrates the need to make hydrogen, using electrolysis, with renewable based energy. |
Hydrogen Economy: Taking energy dependence from fossil fuel to hydrogen
|
The upper part of figure 7 is referred to as the transitional phase. Currently, there is no infrastructure (or economy) that allows hydrogen to be implemented as a fuel. In the beginning, fossil fuels will be used to create hydrogen energy, mainly by the use of Steam Reformation. H2ICE's can run on both hydrogen and gasoline and can act as a "bridging technology" which can "assist with the gradual build up of a hydrogen fueling infrastructure." [3] The bottom end of figure 7 shows the long-term, post fossil fuel age. The plan here is to produce hydrogen from renewable resources and produced using a method like Electrolysis, which is very clean. Information and Image take from [2] unless directly stated. |
Article Sources
[1] Tomorrow's Energy by Peter Hoffmann, 2012
[2] Towards Sustainable Road Transport by Ronald M. Dell, Patrick T. Moseley, David A. J. Rand, 2014
[3] Renewable Hydrogen Technologies by Sebestian Verhelst et al, 2013
[4] Gaines L.L., Elgowainy A., Wang M.Q., Full fuel-cycle comparison of forklift propulsion systems. Argonne National Laboratory.
Supplemental Information from....
[1] Tomorrow's Energy by Peter Hoffmann, 2012
[2] Towards Sustainable Road Transport by Ronald M. Dell, Patrick T. Moseley, David A. J. Rand, 2014
[3] Renewable Hydrogen Technologies by Sebestian Verhelst et al, 2013
[4] Gaines L.L., Elgowainy A., Wang M.Q., Full fuel-cycle comparison of forklift propulsion systems. Argonne National Laboratory.
Supplemental Information from....
- Air Products. (2013). Steam methane reformer. Retrieved from http://www.airproducts.com/~/media/Files/PDF/industries/energy-hydrogen-steam-methane-reformer-datasheet.pdf
- Verhlest, S., Wallner, T. (2009). Hydrogen-fueled Internal Combustion Engines. Progress in Energy and Combustion Science, 35(6), 490-527. doi: 10.1016/j.pecs.2009.08.001